Lab 2a: "Dissecting" a "cell" and Examining Its Components
Purpose: What are positive indicator tests for proteins, carbohydrates, and fats? Do parts of an egg test positive for protein, carbohydrate, and/or fat?
Materials:
Procedure:
Part 1-
Part 2-
Test the egg for monosaccharides, starch, proteins, and lipids using the appropriate tests, recording the changes as you go.
Monosaccharides:
Starch:
Protein:
Lipid:
Part 3-
Now perform these tests on the egg, substituting the egg for the standard solutions (starch, protein, etc.), and record the outcomes, especially color changes.
Results:
Part 1-
Day 1: We take the egg and soak it in acetic acid for 24 hours.
Day 2: After taking the egg out of the vinegar, the egg's shell was almost completely dissolved, with some staying on the egg in the form of a salt-like material. The egg also seemed to diminish in size. We then soaked the egg in a salt solution for a day.
Day 3: After taking out the egg out of the salt solution, we noticed that more of the egg's shell had fallen off (those stubborn few pieces that still clung on after the vinegar), the egg was paler, and there were fewer bubbles in the water. The egg was then put into a beaker of distilled water for another day.
Day 4: Testing day! After taking the egg out of the water, the membrane was even paler, practically transparent, the yolk coloring the egg slightly yellow.
Part 2- (Hit Ctrl + to enlarge)
Materials:
- Eggs
- 250 mL Beaker
- White Vinegar
- Plastic Wrap
- Slotted Spoon
- Sodium Chloride
- Distilled Water
- 100 mL Beaker
- 5 mL Pipets
- Green Pipet Pump
- 13x100 mm Glass Tubes
- Peg Racks for 13x100 mm Tubes
- Glucose (Dextrose)
- Benedict's Solution
- Hot Plate Stirrer
- Test Tube Holder
- Soluble Starch
- Lugol's Iodine Solution
- Vortex Mixer
- Gelatin
- Sodium Hydroxide
- Cupric Sulfate 5-Hydrate
- Oil
- Sudan IV Solution
- #4 Scalpel Handles
- #22 Scalpel Blades for #4 Handles
- Plastic Trays.
Procedure:
Part 1-
- Dissolve egg shell in white vinegar for one to two days
- Use a slotted spoon to pick up the egg and use water to rinse the vinegar off of the egg
- Leave the egg in a 5% NaCl (Sodium Chloride) solution for a day to see if water can permeate the outer membrane.
- Rinse the egg and put it into a beaker of distilled water for a day.
- Cut open the egg and let the egg whites drip into a 100 mL beaker through the slots on the spoon, without breaking the yolk.
- Put the yolk into a different beaker, and save the egg membranes for later.
Part 2-
Test the egg for monosaccharides, starch, proteins, and lipids using the appropriate tests, recording the changes as you go.
Monosaccharides:
- Test for glucose by: mixing 2mL of 2% glucose solution with 2 mL of Benedict's Solution in a test tube. Heat in 100 mL of hot water (100 Degrees C) in a 250 mL beaker for 2 minutes.
- To test for water, use a mixture of 2 mL deionized water and 2 mL of Benedict's sol ution, and repeat the heating step above.
Starch:
- Mix 2 mL of starch suspension (well-mixed) and 0.25 mL of Lugol's iodine. Don't heat, merely swirl in.
- Test for water again, using a mixture of 2 mL deionized water with 0.25 mL of Lugol's iodine.
Protein:
- Put 2mL of gelatin, and 1 mL of Biuret Solution (be sure to wear goggles and gloves), and put on the vortex mixer. Then add 0.25 of 5% copper sulfate, and vortex the mixture again. The mixture should change color within 30 seconds.
- Water test using the NaOH-CuSO4 solution.
Lipid:
- Add 2 mL of oil to 60 uL of Sudan IV, and swirl to mix.S
- Add 2 mL of water to 60 uL of Sudan IV and swirl to mix.
Part 3-
Now perform these tests on the egg, substituting the egg for the standard solutions (starch, protein, etc.), and record the outcomes, especially color changes.
Results:
Part 1-
Day 1: We take the egg and soak it in acetic acid for 24 hours.
Day 2: After taking the egg out of the vinegar, the egg's shell was almost completely dissolved, with some staying on the egg in the form of a salt-like material. The egg also seemed to diminish in size. We then soaked the egg in a salt solution for a day.
Day 3: After taking out the egg out of the salt solution, we noticed that more of the egg's shell had fallen off (those stubborn few pieces that still clung on after the vinegar), the egg was paler, and there were fewer bubbles in the water. The egg was then put into a beaker of distilled water for another day.
Day 4: Testing day! After taking the egg out of the water, the membrane was even paler, practically transparent, the yolk coloring the egg slightly yellow.
Part 2- (Hit Ctrl + to enlarge)
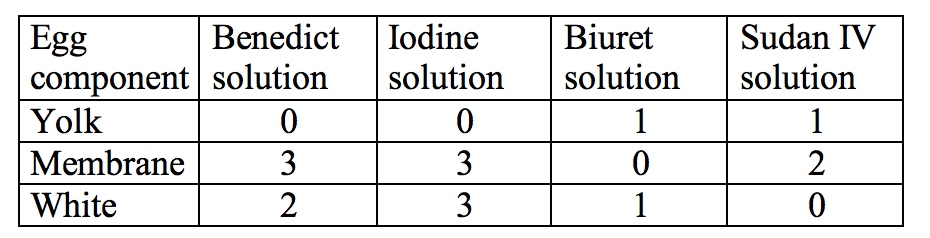
Part 3-
Data Analysis:
After finishing this lab and looking at the results, we were able to answer the purpose question and have evidence to back it up. We found that the positive indicator for protein, carbohydrates, and fats were a blue/purple color, a brown/black color, and a milky red/pink, respectively. There are definitely parts of an egg that test positive for different organic substances; in this case the yolk was positive for protein and fat, the membrane for monosaccharides, iodine and fats, and the egg whites tested positive for everything but fat. In every experiment there is always some error, in this case it would probably be problems with measurement and cross-contamination. We reused the pipets for the same liquid, and some of the labels were rubbed off, so there definitely was a chance that liquids were mixed inadvertently. Also, the egg membrane wasn't accurately measured, merely cut up into four pieces that weren't exactly equal. If I were to do this again, I would use more than one egg to get an average score, and eliminate as many variables as possible. It would also be interesting to see if different types and sizes of eggs (A, AA, free range, etc) would make a difference. Indicator tests could also be used to measure any number of things, from testing to see if certain harmful chemicals were in food, to determining if there were specific proteins missing from a blood sample.
After finishing this lab and looking at the results, we were able to answer the purpose question and have evidence to back it up. We found that the positive indicator for protein, carbohydrates, and fats were a blue/purple color, a brown/black color, and a milky red/pink, respectively. There are definitely parts of an egg that test positive for different organic substances; in this case the yolk was positive for protein and fat, the membrane for monosaccharides, iodine and fats, and the egg whites tested positive for everything but fat. In every experiment there is always some error, in this case it would probably be problems with measurement and cross-contamination. We reused the pipets for the same liquid, and some of the labels were rubbed off, so there definitely was a chance that liquids were mixed inadvertently. Also, the egg membrane wasn't accurately measured, merely cut up into four pieces that weren't exactly equal. If I were to do this again, I would use more than one egg to get an average score, and eliminate as many variables as possible. It would also be interesting to see if different types and sizes of eggs (A, AA, free range, etc) would make a difference. Indicator tests could also be used to measure any number of things, from testing to see if certain harmful chemicals were in food, to determining if there were specific proteins missing from a blood sample.
Reflection: I liked this lab, it was a good introduction to biotech, and mixed biology with one of my favorite division of science, chemistry (but of course, all biology is chemistry, and all chemistry is physics). This was also a very forgiving lab if you screwed up, the very few times that I spilled something or misplaced another, I was back on track within a matter of minutes, and didn't have to start the whole lab over again. I was really happy with my partner's and my collaboration and teamwork, we both gave and took instruction when appropriate (don't cross he who possesses the chemicals), kept a good attitude, didn't clash or quarrel, and just worked well together in general. If I was to do this lab again, I would have probably only used pipets that I knew belonged with which liquid, and would've had fewer mistakes now that I know the lab a little better.